Golden Gophers Fighting COVID-19 Together!
Today we are facing a global health crisis due to the sudden outbreak of the coronavirus where the number of diagnosed cases are exponentially increasing each day, while it still remains uncertain as to how many cases are undiagnosed. The fact that we are still dependent on the traditional PCR technique for its diagnosis, just confirms that the medical device market still lacks an efficient point-of-care (POC) detection device for portable, rapid diagnosis of diseases. We the researchers from the Department of Electrical & Computer Engineering, Center for Infectious Diseases and Microbiology Translational Research (CIDMTR), Department of Medicine, Institute for Molecular Virology, Department of Veterinary Population Medicine (VPM), Department of Mechanical Engineering, and Department of Computer Science & Engineering, under the leadership of Distinguished McKnight University Professor and Robert F Hartmann Chair Prof. Jian-Ping Wang and Associate Professor Maxim C. Cheeran are developing POC devices that have the potential for rapid detection of COVID-19. Over the years, Prof. Wang's Nanospin lab is developing portable testing kits based on magnetic particle spectroscopy (MPS) technology to realize rapid, sensitive, and multiplexed on-site disease detection. In our recently published ACS AMI paper (download link, not for commercial use), we have successfully demonstrated the feasibility of using a MPS system combined with the self-assembly of nanoparticles for rapid, sensitive, and wash-free detection of H1N1 virus.
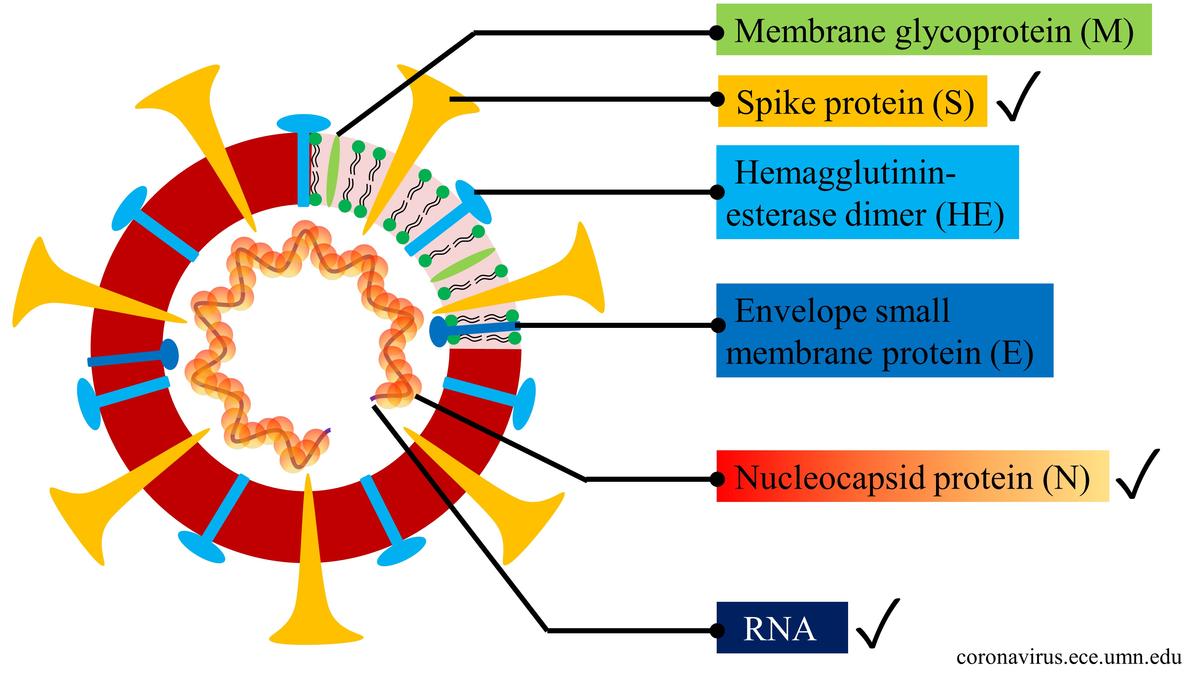
As shown below, the SARS-CoV-2 is enveloped positive sense RNA virus. A virus consists of spike protein (S), hemagglutinin-esterease dimer (HE), membrane glycoprotein (M), envelope protein (E), nucleoclapid protein (N) and RNA. Spike protein (S) mediate attachment to host receptors. It is the largest structure and makes the distinct spikes on the surface of the virus. Nucleocapsid protein (N) binds to viral RNA genome. Envelope protein (E) facilitates assembly and release of the virus.
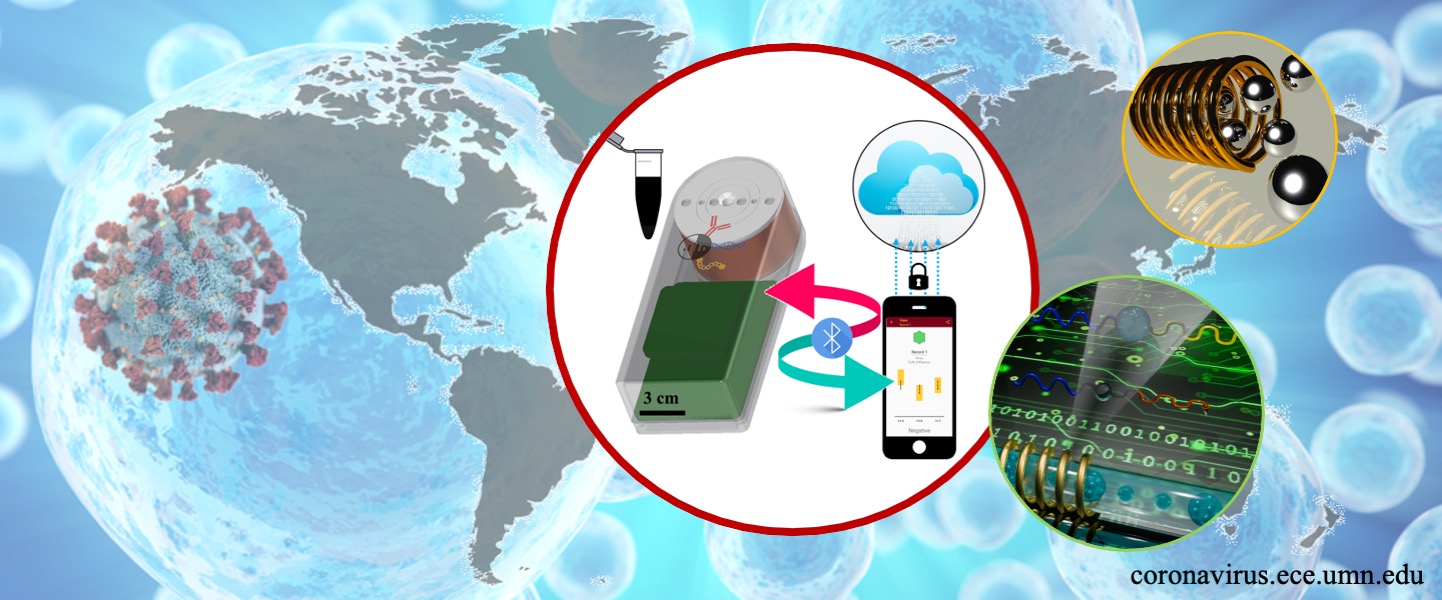
The working principle of the technology could allow the detection of SARS-CoV-2 protein antigens, mRNA, and the antibody response to the virus. These MPS devices coupled with smartphone interfaces will allow testing in remote areas and on-site settings, such as in households and small clinics. By transmitting test results collected from distant locations to centrally located data analysis units, state and federal government can have real-time epidemiological data at their fingertips. This would also significantly reduce the costs for monitoring infections at the regional level. These possibilities would alter current paradigms for disease surveillance and enhance the ability to evaluate and monitor disease control protocols in real-time. It is expected that this new technology will transform today’s expensive and labor-intensive sensing techniques into a user-friendly and cost-effective detection protocol with superior or comparable sensitivity and specificity, which could contribute to the surveillance and control of SARS-CoV-2.
In MPS system, when applying AC magnetic fields to magnetic nanoparticles (MNPs), their magnetic responses can be monitored by a pair of pick-up coils, in the format of harmonics. We conjugate specific antibodies or RNA strands onto MNP surface. In the presence of target biomarkers (such as S and N protein, RNA), MNPs link to biomarkers through specific antibody-antigen (RNA-RNA) interactions. This specific recognition process allows us to detect specific biomarkers from biofluid samples. The magnetic responses of MNPs linked to target biomarkers become weaker. Converting the concentration of biomarkers into magnetic signals and electrical signals from pick-up coils. The figure below shows a schematic view of how our specially designed MNP probes detect the target biomarkers for the detection of SARS-CoV-2.
Please follow this site for any further updates on our MPS technology for COVID-19 detection.
University of Minnesota COVID-19 relief